
AN AUTHORITATIVE RESOURCE FOR EVERYTHING ABOUT CANCER
2026 Updates: What Cancer Doctors Are Saying About Ivermectin and Cancer Treatment

April 2026 Updates: What Cancer Doctors Are Saying About Ivermectin and Cancer Treatment
Is ivermectin a viable cancer treatment? Despite growing online claims and renewed attention from the National Cancer Institute (NCI), oncologists emphasize that evidence remains limited. This updated 2026 survey explores expert perspectives, current research, and the risks of misinformation surrounding ivermectin and cancer care.
Why Ivermectin Is Back in the Cancer Conversation in 2026
The NCI Adopts a Stance—But What Do Doctors Say?
If you've spent time in online cancer communities recently—or if you’ve taken any interest in livestock welfare in the last 50 years—you've almost certainly encountered ivermectin. The antiparasitic drug, long used to treat parasitic infections like river blindness and scabies, has become a fixture in the word-of-mouth medical world. Podcasts, patient forums, social media groups, and clinic waiting rooms are abuzz with the possibility that ivermectin can cure cancer. And in early 2026, the conversation got louder: the National Cancer Institute (NCI) confirmed it is actively studying ivermectin as a potential cancer treatment.
Specifically, the research giant has initiated a preclinical study looking into the anti-tumor efficacy of ivermectin. Preclinical studies are precursors to human clinical trials. This is a critical distinction for patients to consider, according to oncologists who have their patients’ health as their highest priority. According to scientists at the NCI, speaking anonymously to KFF Health News, the action isn’t altogether benign: “We are moving funds away from so much promising research in order to do a preclinical study based on nonscientific ideas. It’s absurd.”
Notably, Anthony Letai, NCI director since September 2025 and proponent of the increased laboratory focus on ivermectin, makes no sweeping promises. “At least on a population level, it’s not going to be a cure-all for cancer,” he said.
Are other oncologists as optimistic—or as pessimistic—about the touted wonder drug? The Cancer News surveyed oncologists to find out. This December 2025 article by The Cancer News has been updated to reflect recent updates in ivermectin research, policy, and public opinion.
From COVID-19 Controversy to Cancer Claims: How Ivermectin Gained Attention
The COVID-19 Pandemic claimed over 7 million lives globally, and another dangerous infection spread alongside it: medical misinformation. At the height of this global event, the conversation around nontraditional cures homed in on the antiparasitic called ivermectin. The drug gained popularity after inconclusive preliminary research, with some people believing it could cure people with COVID-19 infection. After the fact, a 2024 study published in the New England Journal of Medicine found that ivermectin had no meaningful benefit in the treatment of COVID-19 infection, and articulated concerns about the quality of previous studies that had found a benefit. WHO concluded that “there existed only very-low-certainty evidence regarding ivermectin.”
Still, the drug’s reputation outpaced the evidence. Now, that reputation is gaining traction again in the field of cancer. In a poll conducted by The Cancer News, more than 90% of medical oncology professionals indicated they had been asked by patients or caregivers about using ivermectin for cancer treatment. "There is too much misinformation out there," said one oncologist. To address this concern and clarify what the evidence actually shows, we asked oncologists to share their professional perspectives. But first, what is ivermectin, and what is it used for?
What Is Ivermectin? Uses, History, and FDA Approval
The story of ivermectin began in the 1970s near a golf course in Honshu, Japan. The microbiologist Satoshi Ōmura collected soil samples from the woods and went back to the laboratory for analysis. There, he discovered a new bacterium, Streptomyces avermitilis, which showed strong activity against parasitic infections in mice. From this, Omura purified the active components, revealing a family of compounds called avermectins, and by refining them, ivermectin was developed.
Ivermectin is a drug with powerful antiparasitic properties effective against both animals and humans. In the 1980s, it became one of the highest-selling animal health products due to its potency against both endoparasites (those that live in the body) and ectoparasites (those that live on the surface of the body). Ivermectin works by blocking ion channels in parasites and invertebrates, causing paralysis. This paralysis disrupts a parasite’s neuromuscular function, ultimately causing its death.
Currently, the ivermectin tablet is approved by the FDA to treat people with a specific roundworm infection and river blindness. The topical version of ivermectin is approved for external parasites such as head lice and rosacea. The FDA has cautioned against the use of Ivermectin in conditions that have not been approved or authorized. While for COVID-19, clinical trials demonstrate ineffectiveness of ivermectin, for cancer, there are limited data supporting ivermectin’s efficacy. There is only one active phase 1/2 clinical trial testing the combination of ivermectin and two immunotherapy drugs for patients with metastatic triple-negative breast cancer (TNBC). The trial is expected to finish by 2026, but if the results look promising, it will still need phase 3 testing. As of May 2026, results are pending.
Do Oncologists Recommend Ivermectin for Cancer Treatment?
We’re left with two questions, then. For one, what exactly do we know about ivermectin’s efficacy against cancer? And two, what should we—patients, caregivers, and the general public—do with that information as clinical trials progress? We asked oncologists to weigh in on both fronts.
It is important to distinguish that we know very little while we lack conclusive phase 3 trial results. “There is extremely limited data that ivermectin has a role in cancer biology,” Dr. Adeel Khan, hematologic oncologist and epidemiologist at the University of Texas Southwestern Medical Center, told The Cancer News. “Some in vitro studies suggest antiproliferative properties or pro-apoptotic properties, but this data is small-scale and early, and not yet convincing,” he added.
Antiproliferative properties inhibit cancer cell division and growth, while pro-apoptotic properties actively induce programmed cell death; in other words, one stops "more" cancer cells from forming, and the other eliminates "current" cancer cells, respectively.
Dr. Siddhartha Yadav, an oncologist at Mayo Clinic, put in another bid for caution. “It is critically important that we prove or disprove the efficacy of ivermectin in the management of cancer so that we, the clinicians, are able to provide evidence-based recommendations, and patients can make informed decisions about its role in cancer [treatment].”
Current research in ivermectin leaves room for that role to range from nonexistent to negligible to transformative. The key takeaway from both hem/oncologists: All we conclusively know on the subject is that further research is needed. Proceed with caution if you run into a source claiming conclusivity without the medical evidence to back it up.
That leaves us with the second question. How should this update impact treatment in the here-and-now?
“The data is simply lacking for patients to take ivermectin as a bona fide chemotherapeutic agent, especially in lieu of conventional, evidence-based therapies,” Khan stressed. Provided the NCI’s ivermectin study follows ethical guidelines, Khan is supportive. “However, we as a scientific community will review the forthcoming data with a fine-tooth comb.”
“At the end of the day, our goal is to recommend and prescribe treatments that serve the patient’s best interests and we are guided consistently by the available scientific evidence in that regard,” Yadav added.
Adeel Khan, MD, MPH, MS is a hemoncologist and epidemiologist at the University of Texas Southwestern Medical Center and notes his views do not necessarily represent his employer.
Siddhartha Yadav, MBBS., MD, is a medical oncologist specializing in treatment of breast and gynecologic cancers at Mayo Clinic.
Survey Insights: Why Patients Ask About Ivermectin in Cancer Care
Claims about ivermectin as a cancer treatment have been circulating in the media since 2020. These claims have reached the clinic, and oncologists are expressing dissent, skepticism, and frustration. We surveyed a sample of 30 oncologists on their point-of-care exposure to ivermectin.
Context of Patient Requests for Ivermectin for Cancer Treatment in Hospital Settings
Oncology healthcare professionals described the situations in which patients with cancer raise the topic of ivermectin. Most oncologists report that this happens after patients read information online or on social media, often from non-medical sources. Some patients bring it up, hoping to enhance the effectiveness of their care, suggesting it as an adjunct or alternative to ongoing treatment. These findings underscore the importance of patient education on cancer treatment and evidence-based medicine. Providers play a key role in ensuring that patients make well-informed decisions to counter preexisting notions that may be inconclusive.
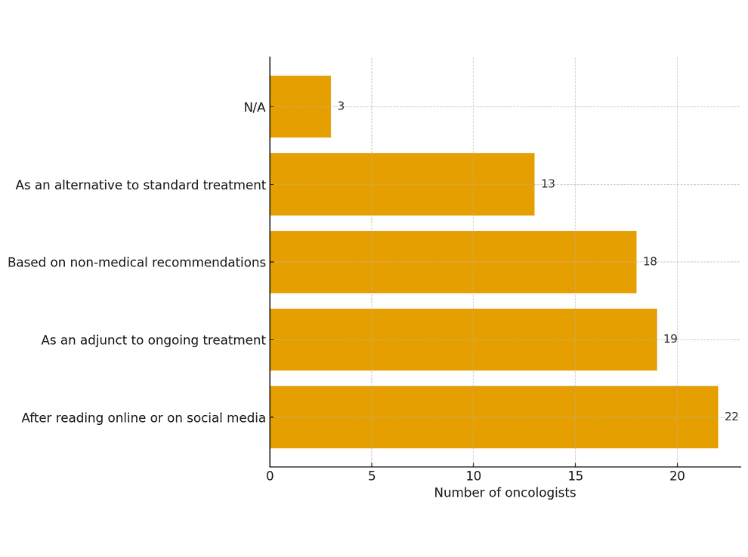
How Oncologists Respond to Ivermectin Requests
Echoing the scientific literature, the vast majority of oncology professionals cite the lack of conclusive evidence whenever patients request ivermectin for cancer treatment. Other oncologists strongly discourage its use, avoid the question, or refer patients to existing clinical studies.
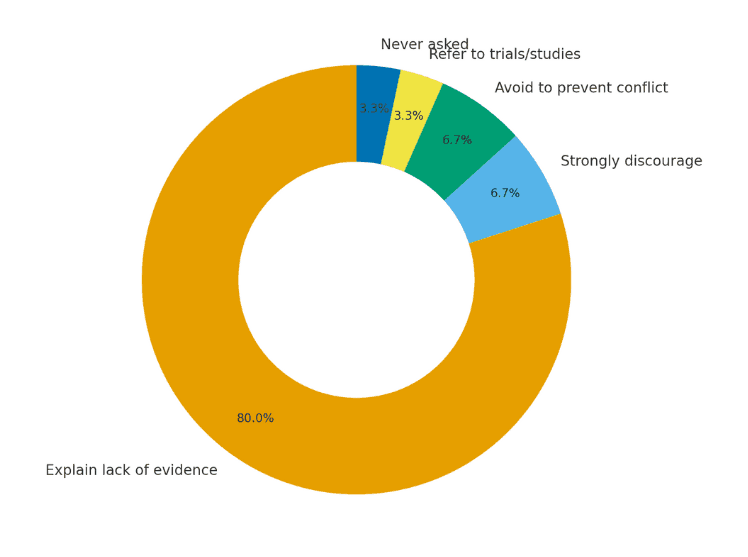
Reported Risks and Clinical Concerns
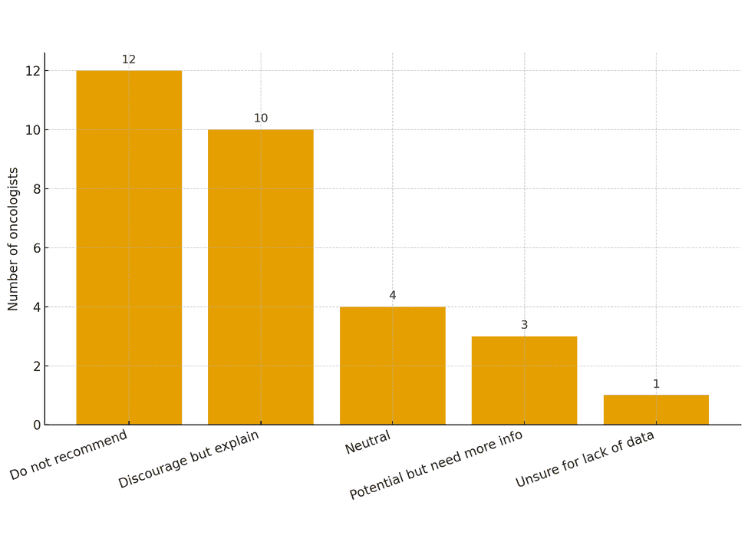
Several of the oncologists we surveyed do not recommend ivermectin for cancer treatment. One oncologist recounted a specific scenario that illustrates the dangers of using unapproved medication, sharing, “In those patients who did not admit to using it (until intubated and spouse mentioned it or in one case where I pushed ) they had had toxicity that we attributed to chemo but which, in retrospect, was likely ivermectin induced.” Another oncologist also mentioned that “I had a patient taking this while not accepting standard cancer care, and cancer progressed.” These responses further underscore the need to raise awareness about the cancer research process and why drugs go through rigorous testing before approval.
Awareness of Ivermectin Clinical Trials Among Oncologists
While most oncologists are not aware of any ivermectin clinical trials, one shared: “There was a plan for a phase II ivermectin/immunotherapy trial in triple-negative breast cancer that was halted/pulled, and a currently recruiting trial on clinicaltrials.gov for the same in TNBC MBC. So, there is some preclinical data suggesting it may be effective, but this is not the Phase III RCT clinical trial data we use in most of our studies.”
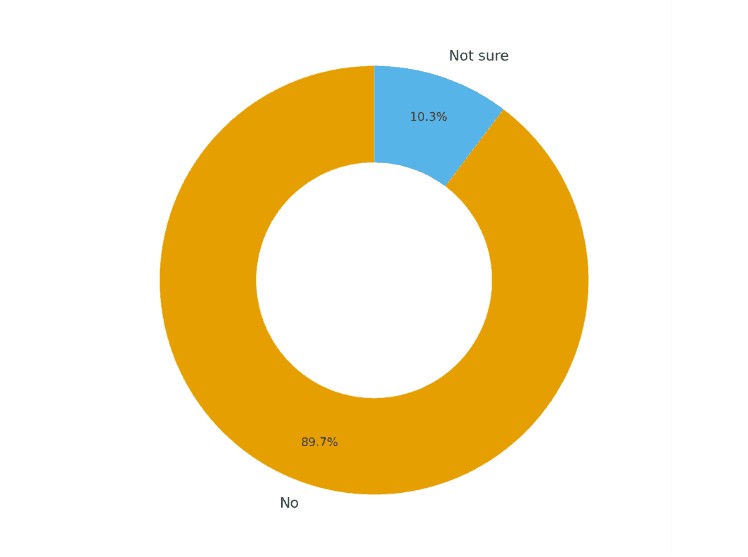
The doctor's response highlights that the use of ivermectin for cancer treatment is largely predicated on preclinical studies, which may not translate well to humans. As such, it is important to await robust data from a Phase III clinical trial before drawing any definitive conclusions.
Current Evidence on Ivermectin and Cancer: What Patients Should Know
As of April 2026, the NCI has initiated preclinical trials on ivermectin’s efficacy relating to cancer. No updates on the progress of the trials are available, and the conclusions of this survey have not changed.
There are over 20,000 scientific publications about ivermectin and cancer. The majority of these publications are preclinical, meaning the results show that ivermectin has an anticancer effect at the cellular level—in cells plated in a glass dish or implanted in mice, all of which is completely outside the human body. It does show some promise in the lab, but there is currently no proven clinical role for ivermectin in cancer treatment. In our survey, some oncologists agreed that more clinical trials are needed before it could ever become part of standard care. It is important for patients to openly discuss their concerns about ivermectin with their doctors and ask about the latest clinical trials to make well-informed decisions.
Frequently Asked Questions - Ivermectin and Cancer: What Patients Need to Know (Updated April 2026)
What is ivermectin?
Ivermectin is a medication approved to treat parasitic infections like river blindness and head lice. It is not currently approved to treat cancer.
Why are people talking about it for cancer?
Lab studies (done in dishes and in mice) have shown ivermectin may have some effect against cancer cells. This has fueled widespread interest online and in patient communities. But lab results do not always translate to effects in the human body.
What is the NCI doing?
The National Cancer Institute (NCI) launched a preclinical study in early 2026 to investigate ivermectin's potential against cancer. Preclinical means it is still in the early research phase — not yet tested in human clinical trials at scale.
What do oncologists (cancer doctors) say?
The vast majority do not recommend ivermectin for cancer treatment. Their main concerns are the lack of solid human trial data, potential drug interactions and toxicity risks, and cases where patients who used it without telling their doctors experienced harm.
Is there any human trial data?
Yes — there is one active early-phase clinical trial (Phase 1/2, NCT05318469) at Cedars-Sinai Medical Center testing ivermectin combined with immunotherapy drugs for metastatic triple-negative breast cancer. Of 8 evaluable patients, 6 had progressive disease. Two patients showed some benefit, but because ivermectin was given alongside approved immunotherapy drugs, it is difficult to determine whether any benefit was due to ivermectin itself or the immunotherapy drugs it was combined with. Full results are expected by late 2026, and even if promising, a much larger Phase 3 trial would still be needed before this could become a standard treatment.
Understanding clinical trial phases — and why they matter for ivermectin
Clinical trials follow a stepped process designed to protect patients and ensure treatments are both safe and effective before doctors can responsibly recommend them:
- Phase 1 tests a drug in a small group of people — often fewer than 30 — primarily to assess safety, identify side effects, and determine safe dosing. It does not tell us whether the drug works.
- Phase 2 expands to a larger group, typically up to a few hundred patients, to get early signals of whether the drug may be effective and to continue evaluating safety.
- Phase 1/2 (where ivermectin currently is) combines both of these early steps — it is still primarily a safety and dose-finding exercise with only preliminary hints at effectiveness.
- Phase 3 is the gold standard. It involves hundreds to thousands of patients across multiple sites, compared against existing treatments or a placebo, in a rigorously controlled setting. This is the level of evidence oncologists need before they can safely and confidently recommend a treatment.
Ivermectin is at the very beginning of the human testing journey. For context, most drugs that enter Phase 1 never make it to Phase 3 — and many that reach Phase 3 still fail. Until ivermectin completes a successful Phase 3 trial in cancer patients, there is simply not enough evidence for oncologists to recommend it as part of cancer care.
What are the risks of taking ivermectin — especially without telling your care team?
Ivermectin is not harmless just because it is an approved drug for other conditions. Taking it during cancer treatment carries real risks:
- Drug interactions: Ivermectin can interact with chemotherapy and immunotherapy drugs in ways that are not yet fully understood, potentially making your cancer treatment less effective or increasing side effects.
- Toxicity: At higher doses, ivermectin can cause nausea, vomiting, diarrhea, dizziness, low blood pressure, muscle weakness, and in severe cases, seizures, coma, or death.
- Masking side effects: If you experience symptoms while taking ivermectin alongside cancer treatment, your care team may attribute them to your cancer drugs and adjust your treatment unnecessarily — potentially putting you at greater risk.
- Delayed or abandoned standard care: Some patients have chosen ivermectin over proven treatments. Oncologists have reported cases where patients who did this experienced cancer progression that may have been preventable.
- Hidden use is dangerous: If you are taking or considering ivermectin, tell your oncologist. They are not there to judge you — they need the full picture to keep you safe.
The bottom line Ivermectin shows some early promise in the lab and is being studied in humans for the first time — but the evidence is still very limited and far from conclusive. The responsible path forward is more research, not widespread use. If you have questions about ivermectin or any other supplement or medication, talk openly with your oncologist before taking anything outside of your prescribed treatment plan.
Works Discussed
- Laing, R., Gillan, V., & Devaney, E. (2017). Ivermectin - Old Drug, New Tricks?. Trends in parasitology, 33(6), 463–472. https://doi.org/10.1016/j.pt.2017.02.004
- Sai Yengu, N., Raheem, A., Pons, A. G., Ho, W. L., Ali, S. M. S., Haseeb, A., Fadlalla Ahmad, T. K., & Mustafa, M. S. (2025). The impact of ivermectin on COVID-19 outcomes: a systematic review and meta-analysis. Annals of medicine and surgery (2012), 87(2), 809–829. https://doi.org/10.1097/MS9.0000000000002762
- U.S. Food and Drug Administration. (2024, April 5). Ivermectin and COVID-19. https://www.fda.gov/consumers/consumer-updates/ivermectin-and-covid-19
- Pradhan R. US cancer institute studying ivermectin's 'ability to kill cancer cells.' KFF Health News. February 10, 2026. Accessed April 20, 2026. https://kffhealthnews.org/news/article/ivermectin-cancer-treatment-nih-study-dewormer-offlabel-drug/
Author
The Cancer News Team
Categories
Share Article





